Lim, Park, Lee, and Park: Changes in Heart Rate, Muscle Temperature, Blood Lactate Concentration, Blood Pressure, and Fatigue Perception Following Jogging and Running: An Observational Study
Abstract
Purpose
To report the magnitude of changes in heart rate, muscle temperature, blood lactate concentration, blood pressure, and fatigue perception after treadmill jogging and running.
Methods
Eleven healthy male adults (174±7 cm, 72±10 kg) visited the laboratory twice. After a 10-minute rest each visit, subjects performed a treadmill jog (maintaining a speed of 9 km/h at a 1% incline) or a treadmill run (initially 5 km/h at a 1% incline, belt speed, and incline increased by 1 km/h and 0.5% every minute; average speed of 9 km/h). Heart rate and muscle (gastrocnemius) temperature were recorded before, during, and after the treadmill jog/run. Blood lactate concentration, blood pressure, and fatigue perception were assessed before and after. The effect of the condition over time was analysed using parametric or nonparametric tests (p≤.05) with Cohen's d effect sizes.
Results
There was a condition effect over time in heart rate (F1,30 =22.16, p<.0001), blood lactate concentration (χ2 =34.88, df=3, p<.0001), systolic blood pressure (F1,30 =4.18, p=.05), and fatigue perception (F1,30 =10.24, p=.003). Specifically, subjects who ran showed a higher heart rate (187 vs. 158 bpm, p<.0001, d=2.11), blood lactate concentration (11.4 vs. 3.8 mmol/L, p<.0001, d=3.39), systolic blood pressure (164 vs. 147 mmHg, p=.006, d=1.34), and fatigue perception (2.7 vs. 5.0 cm, p=.0005, d=1.46) than those who jogged. There was a time effect (F1,30=94.84, p<.0001) on muscle temperature, with a 1.5°C increase in muscle temperature after jogging/running (p<.0001, d=1.88).
Conclusions
While treadmill jogging and running induced a similar temperature increase in the gastrocnemius, running resulted in higher heart rate, blood lactate concentration, systolic blood pressure, and fatigue perception. These results can be used as a basis for planning warm-up protocols.
Keywords: Warm-up, Physiological Responses, Treadmill Run, Gastrocnemius Temperature
INTRODUCTION
A warm-up is practiced for explosive muscle contractions and strenuous joint movements during main exercises. Among those in the field, either jogging or running is a most common form of warm-up activity. Jogging and running are differentiated from walking as they include a flight phase [ 1]. Jogging and running can be further distinguished by the vertical ground reaction force traces: the peak values are observed at midstance during running while it is not during jogging [ 2]. In the field, however, this type of kinetic distinction between jogging and running is unattainable. Therefore, the pace (e.g., the speed at which an individual moves) is a typical determinative factor. Previously, the threshold of jogging speed was reported as 9 km/h [ 3] or 10 km/h [ 4]. There are pros and cons of each warm-up mode. Jogging requires a low energy expenditure relative to running [ 5]; thus, each mode utilises different energy systems when completed within the same duration. For example, an individual who jogs could maintain the oxidative energy system when performing at a constant speed, but an individual who runs could probably be on the glycolytic energy system. Due to a higher intensity, running is thought to be more efficient to raise the core and muscle temperature relative to jogging, but early energy exhaustion could occur, which will be an obvious disadvantage for overtime. It is important to know cardiovascular response, muscle temperature, and physiological and psychological fatigue when planning and determining the parameters of warm-up methods (e.g., mode, duration, and intensity). Up to date, there have been numerous studies [ 6– 10] reporting the changes following various warming-up on aforementioned outcomes. Although the data from these studies are valuable, the body's immediate responses following the most popular modes of warm-up, jogging and running, are not fully understood. A recent systematic review suggested that a period of 10-minute warm-up appears to be effective in maximising best athletic performance [ 11]. A direct comparison between a 10-minute jogging and running would help us to characterise each mode of warm-up in terms of cardiovascular response, muscle temperature, and physiological and psychological fatigue. Strength and conditioning personnel would benefit from knowing the status or level of an individual who performs jogging or running. Athletes would further benefit from having options when choosing their best warm-up strategy. Therefore, we were interested in quantifying the magnitude of changes in heart rate, the gastrocnemius temperature, blood lactate concentration, blood pressure, and fatigue perception following treadmill jogging or running. While we expected that subjects with the running condition would show higher values in each dependent measurement than those with jogging. Due to the intensity, individuals who perform similar exercise as warm-up would benefit from referencing the results when intervening warm-up activities at the similar duration and intensities.
METHODS
1. Design Overview and Experimental Approach
A repeated-measures design was used in which heart rate, muscle temperature, blood lactate concentration, blood pressure, and fatigue perception were recorded during two separate sessions at least two-weeks apart. Subjects were asked to keep their habitual diet and fast within three hours prior to visiting the laboratory. As mentioned in the introduction, the speed of jogging was defined as <9 km/h [ 3] or 10 km/ h [ 4]. The study concerning about the peak vertical ground reaction force to define jogging reported a speed of 7.2 km/h [ 2]. This suggests that 7.3 km/h could be considered as running. After conducting a pilot study on a treadmill for jogging speeds, we decided that a belt speed of 7.3 km/h was too slow to be a running speed. Similarly, a speed of 10 km/h was too fast for our subjects to complete the 10-minute run. Therefore, we selected a belt speed of 9 km/h for the jogging condition. In each session, subjects performed a 10-minute treadmill jog (a constant belt speed at km/h at a 1% incline) or run (starting at 5 km/h at a 1% incline, then a belt speed of 1 km/h and an incline of 0.5% were increased every minute). The order of exercise protocols was not randomised: all subjects performed treadmill jogging first followed by treadmill running. Mean ambient temperature and relative humidity (standard deviation) in the laboratory were 23.1 (0.6)°C and 43.5 (6.8)% for jogging and 22.8 (0.7)°C and 71.3 (4.4)% for running, respectively.
2. Subjects
Recreationally active, healthy adults who self-reported exercising at least three times per week for at least 30-minute each time were recruited through convenient sampling. Subjects had to be free of any musculoskeletal condition within the last six months, or any cardiovascular or neurological condition in their lives. Eleven individuals (22±2 years, 174±7 cm, 72±10 kg) volunteered for this study. The testing procedures of this study were approved by the institutional review board, and each subject gave written informed consent prior to participation.
The number of subjects were determined by our pilot work. When expecting the difference of 25 bpm (with a standard deviation of 23) in a comparison of postexercise heart rate, a minimum number of subjects to statistically differentiate the treadmill jog and run (with an alpha of 0.05 and a beta of 0.2) was estimated as nine.
3. Testing Procedures
On arrival to the laboratory, subjects laid in a prone position on a treatment table and rested for 10-minute to achieve stability of the autonomic nervous system [ 7] and to attach the heart rate monitor and temperature probe. While in this position, a heart rate monitor (sampled at 60 Hz; Polar Beat, Polar Electro Inc., New York, USA) was applied to the chest. The subjects'sex, height, and weight were entered on the application (Polar H10, Polar Electro Inc., New York, USA), which was wirelessly connected into the monitor, recoding the heart rate values. A thermistor probe (sampled at 60 Hz), connected to a digital logger thermometer (N543, NT logger, NKTC, Tokyo, Japan), was attached to the proximal 1/3 of the medial gastrocnemius on the left limb. Specifically, the thermistor probe was covered by a passive insulation disk (wetsuit material neoprene rubber, size of a 2.5 cm diameter with a 3-mm thick) [ 12], then secured with a film dressing (Tegaderm film, 3M, St. Paul, USA). This technique [ 13] allowed us to noninvasively estimate tissue temperature. The chest strap and the thermistor probe remained in place for the reminder of the 10-minute rest period. At the completion of the rest period, the values in heart rate and gastrocnemius temperature were recorded as the pre-measurements. After-wards, subjects were asked to sit on a chair to obtain pre-measurement values for the rest of the dependent measurements. To assess blood lactate concentration, the subjects’ fingertips were poked by a lancet needle (26G, Moa Lab, Seoul, Korea), then an amount of 0.7 μL was sampled on the test strip (Lactate Test Strips, Nova Biomedical, Waltham, USA). The sampled strip was inserted into the blood lactate concentration analyser (Lactate Plus, Nova Biomedical, Waltham, USA). The value of blood lactate concentration on the monitor of the analyser was recorded [ 14]. Fatigue perception was recorded using a 10-cm visual analogue scale (VAS) [ 9]. The terms “ non-fatigued” and “ fully fatigued” were written at each end of the scale. Subjects were asked to mark a vertical line on the scale to quantify the current level of fatigue. Blood pressure was measured using an electric automated blood pressure monitor (HEM-7121, Omron Healthcare, Kyoto, Japan) [ 15]. While subjects sat on a chair with the left arm resting on the treatment table, an inflatable pressure-cuff (22 cm×32 cm) was wrapped around the brachial artery of the left arm and gently tightened using the Velcro on the pressure-cuff. After pressing the start button, the pressure-cuff was automatically inflated and slowly deflated, the values of the systolic and diastolic blood pressure on the monitor were recorded. Subjects then performed either treadmill jogging or running (see 1. Design overview and experimental approach) at each session. After jogging or running, post-measurements were obtained in the same manner as the pre-measurements.
4. Statistical Analysis
Normal distribution on the baseline at each condition was tested using the Shapiro-Wilk test. If the data were normally distributed, the equality of variance between conditions was then checked using a F-test. If the data showed equal variance, parametric tests were performed. A mixed model (random variable: subject; fixed variables: condition and time) analysis of variance and Tukey-Kramer comparisons as post-hoc tests were conducted. If the baseline data were not normally distributed or the variances were not equal, nonparametric tests (Kruskal-Wallis and Dunn multiple comparisons as post-hoc tests) were performed. The statistical package R (version 4.0.5. R development core team) was used for all tests ( p ≤0.05 for all tests). To determine practical significance, Cohen's d effect sizes (ES=[ X1− X2]/σpooled) [ 16] were also calculated.
RESULTS
Baseline data of all dependent variables were normally distributed, and showed equal variance, except blood lactate concentration (F=8.69, p <.0001, Table 1). Therefore, blood lactate concentration was analysed using nonparametric tests.
Table 1.
Changes in heart rate, gastocnemius temperature, blood lactate concentration, blood pressure, and fatigue perception before and after the treadmill jogging and running
|
|
Jog |
Run |
Statistical Results |
|
Heart rate (bpm) |
|
|
|
|
Pre-measurement |
68.9 (6.3) |
69.2 (6.0) |
W=0.97, p=.87; W=0.96, p=.75; |
|
Post-measurement |
157.7 (17.8)a
|
187.3 (8.7)b,c
|
F=1.66, p=.26; |
|
|
|
|
F1,30 =22.2, p<.0001 |
|
Gastrocnemius temperature (°C) |
|
|
|
|
Pre-measurement |
33.0 (0.7) |
32.9 (0.7) |
W=0.94, p=.51; W=0.96, p=.80; |
|
Post-measurement |
34.7 (0.4) |
34.4 (0.9) |
F=1.16, p=.75; |
|
|
|
|
F1,30 =0.37, p=.55 |
|
Blood lactate concentration (mmol/L) |
|
|
|
|
Pre-measurement |
1.4 (0.4) |
1.3 (0.3) |
W=0.95, p=.61; W=0.95, p=.70; |
|
Post-measurement |
3.8 (2.0) a
|
11.4 (2.5)b,c
|
F=8.69, p<.0001; |
|
|
|
|
χ2 =34.9, df=3, p<.0001 |
|
Systolic blood pressure (mmHg) |
|
|
|
|
Pre-measurement |
127.5 (9.3) |
130.8 (9.9) |
W=0.97, p=.85; W=0.91, p=.27; |
|
Post-measurement |
146.6 (13.0)a
|
163.5 (12.2)b,c
|
F=1.84, p=.20; |
|
|
|
|
F1,30 =4.18, p=.05 |
|
Diastolic blood pressure (mmHg) |
|
|
|
|
Pre-measurement |
77.5 (7.6) |
74.5 (9.4) |
W=0.96, p=.83; W=0.97, p=.88; |
|
Post-measurement |
82.9 (10.1) |
82.6 (6.6) |
F=0.95, p=.91; |
|
|
|
|
F1,30 =0.42, p=.52 |
|
Fatigue perception (cm) |
|
|
|
|
Pre-measurement |
2.7 (1.7) |
3.1 (1.5) |
W=0.90, p=.15; W=0.88, p=.09; |
|
Post-measurement |
2.7 (1.3) |
5.0 (1.7) b,c
|
F=1.55, p=.32; |
|
|
|
|
F1,30 =10.24, p=.003 |
1. Heart Rate
There was a condition effect over time in heart rate (condition×time: F 1,30 =22.16, p <.0001, condition: F 1,30 =23.00, p <.0001; time: F 1,30 = 1,107.30, p <.0001: Fig. 1A). Specifically, heart rate was increased after either condition (jog: p <.0001, d=6.65; run: p <.0001, d=15.75). At post-measurements, subjects who ran showed 19% higher heart rate (187 bpm) than those who jogged (158 bpm, p <.0001, d=2.11).
Fig. 1
Fig. 1Changes in heart rate (A) and gastrocnemius temperature (B) during the treadmill jogging and running. Data are mean (standard deviation). 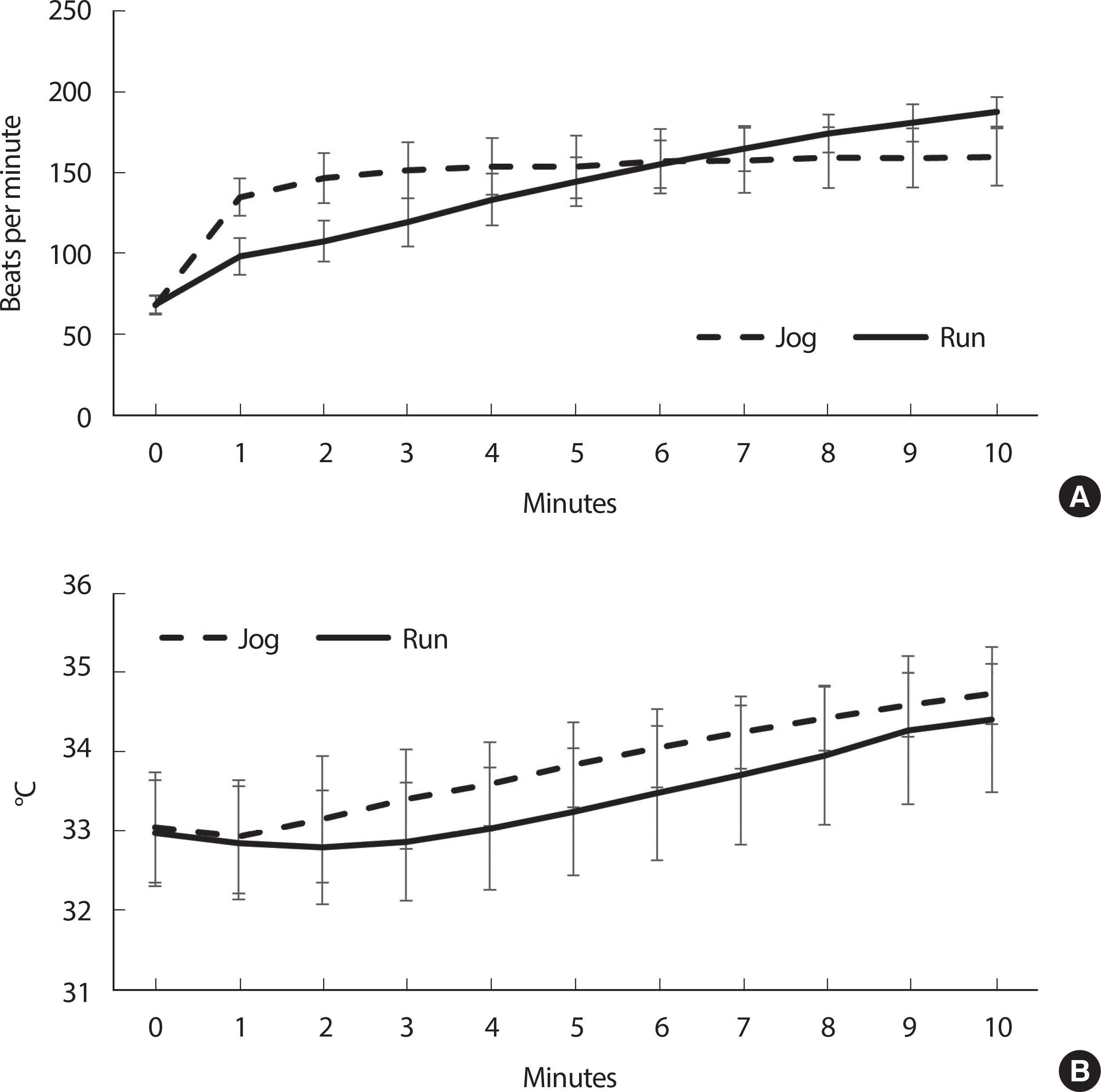
2. Muscle Temperature
There was no condition effect over time in the gastrocnemius temperature (condition×time: F 1,30 =0.37, p =.55, condition: F 1,30 =2.02, p =.17: Fig. 1B). Regardless of condition (time: F 1,30 =94.84, p <.0001), there was a 1.5° C of gastrocnemius temperature increase at post-measurements ( p <.0001, d=1.88).
3. Blood Lactate Concentration
There was a condition effect over time in blood lactate concentration χ 2 =34.88, df =3, p <.0001: Table 1). Specifically, blood lactate concentration increased after jogging ( p =.02, d=1.63) and running ( p <.0001, d= 3.39). However, the amount of increase in running (11.4 mmol/L) was much higher than jogging (3.8 mmol/L, p =.04, d=4.29).
4. Blood Pressure
There was a condition effect over time in systolic blood pressure (condition×time: F 1,30 =4.18, p =.05; condition: F 1,30 =9.15, p =.005; time: F 1,30 =60.31, p <.0001: Table 1) in that the systolic blood pressure increased after the jogging ( p =.02, d=1.70) and running ( p <.0001, d= 2.94). Additionally, a greater systolic blood pressure was recorded after running (131 to 164 mmHg) as compared with the jogging condition (128 to 147 mmHg, p =.006, d=1.34). There was no condition effect over time in diastolic blood pressure (condition×time: F1,30 =0.42, p =.52; condition: F1,30 =0.60, p =.45). Regardless of condition (time: F1,30 =10.41, p =.003), the diastolic blood pressure increased at post-measurements (p =.003, d=0.03).
5. Fatigue Perception
There was a condition effect over time in fatigue perception (condition ×time: F 1,30 =10.24, p =.003, condition: F 1,30 =21.56, p<.0001; time: F 1,30 = 10.24, p =.003: Table 1). There was no jogging induced fatigue (2.7 in VAS for both pre- and post-measurements). However, subjects with the running condition felt 83% greater fatigue perception at post-measurements (3.1 to 5.0 in VAS, p =.0005, d=1.46).
DISCUSSION
The purpose of this study was to compare the body's immediate responses after a treadmill jog and run, and to compare the magnitude in changes. Our hypotheses were mostly accepted in that the running condition resulted in higher values in heart rate, blood lactate concentration, blood pressure, and fatigue perception. These results support the general idea that the intensity of a warm-up protocol is linearly related to physiological and psychological demands. Although they are not a suggestion of a new idea, our data provide the extent of changes in physiological (blood lactate concentration and blood pressure) and psychological (fatigue perception) factors after a short bout of jogging or running. Changes in heart rate or lower-leg muscle temperature recorded in minutes also show our body's immediate response during each exercise. These practical implications could be applied when planning and implementing warm-up protocols prior to exercise training or cardiovascular exercise for rehabilitation.
Cardiovascular activity responds to the demands placed on exercise, which is regulated by the autonomic nervous system [ 17]. Specifically, the heart rate increase during exercise is determined by the facilitation of sympathetic activity and the inhibition of parasympathetic activity [ 18]. At the post-measurement, our subjects showed a higher heart rate when running (187 bpm), as compared with jogging (158 bpm). For the running condition, heart rate was increased linearly with the intensity (treadmill belt speed and incline). Subjects with the jogging condition exercised at a constant intensity (a belt speed of km/h at an incline of 1%). While the heart rate at the end of the jog was recorded as 158 bpm, more than 90% of it was achieved within the first 2-minute of jogging ( Fig. 1A). The rapid increase at the initial stage of jogging (first 2- or 3-minute) is thought to be from the physical demands for the exercise intensity. Although the level of blood lactate concentration and fatigue perception were different, the average values during jogging and running were 143 vs. 141 bpm, respectively. Based on those, we assume that subjects for both conditions spent a similar amount of energy. Since energy expenditure to complete a warm-up activity influences later tasks in athletic performance [ 19], future study should attempt to compare energy expenditure on both warm-up activities. Regarding the amount of increased gastrocnemius temperature, there was a similar amount of temperature increase after the two conditions (jog: 1.7°C; run: 1.5°C). Since a rise in muscle temperature could facilitate the contractile function (e.g., force/velocity relationship) [ 20], our 10-minute warm-up protocols, either jogging, or running, are considered as effective in terms of thermal effects. A recent study [ 9] reported an increase of quadriceps muscle temperature of 1.5 and 2.2°C after the high (80% VO 2max) and moderate (60% VO 2max) intensity of treadmill run, respectively. In this study [ 9], the difference of 0.7°C resulted in a 1 cm difference in vertical jump height. A review paper [ 21] also suggested that a difference in muscle temperature of 1°C could result in the variation of athletic performance by up to 5%. Assuming the other contributing factors are similar (e.g., environmental condition), our exercise conditions would produce a similar level of warm-up effects. We used the insulation disk technique to record muscle temperature, which measures at depth of 2.2 cm from the skin surface of the gastrocnemius [ 22]. This suggests that muscle temperatures at deeper tissues were probably higher than 1.5°C after jogging or running. With that being said, we assume that temperature in the quadriceps muscle was also higher than 1.5°C since it has bigger muscle mass and greater blood flow [ 23, 24]. Although our jogging and running protocol produced similar thermal effects, the advantages related to non-thermal effects (also known as preconditioning the muscles) [ 25] such as postactivation potentiation [ 26], spinal-reflex excitability [ 10], and twitch contraction [ 27] on each warm-up condition could be different. Change in blood lactate concentration is considered as description of the fatigue process, especially in muscle metabolism [ 28]. The blood concentration value after running (11.4 mmol/L) was 67% higher than jogging (3.8 mmol/L). Although not measured, this difference could have been attributed to different energy systems for each condition (e.g., jog: aerobic; run: glycolytic). This is indirectly supported by the changing pattern in other data since the level of heart rate (16%), systolic blood pressure (10%), and fatigue perception (46%) were also higher in subjects with the treadmill run. While a value of the blood lactate concentration >10 mmol/L is considered as the status of severe lactic acidosis [ 29], our running protocol should be considered as a high intensity exercise. Our blood lactate concentration value after running (11.4 mmol/L) could be compared to previous data (11.6 mmol/L) after two maximal 200 m freestyle swimming trials [ 6]. It has been suggested that a variation of 0.7 mmol/L could be considered measurement error at the level of 10 mmol/L when using a portable lactate analyser [ 30]. Since our value (11.4 mmol/L) exceeds the threshold (10 mmol/L), coaches and athletes should be aware of it when interpretating our results. As expected, the systolic blood pressure increased in both conditions (jog: 147 mmHg; run: 164 mmHg) while the diastolic blood pressure did not change over time. Due to differences in the exercise intensity, subjects with the running condition probably needed more oxygen delivery to their working muscles (e.g., lower-extremity). This put an additional demand on the cardiovascular system, which increased the systolic blood pressure and heart rate in our results. While the systolic blood pressure could go up to 190 mmHg (females) or 210 mmHg (males) when exercising at a high intensity [ 31, 32], our value after running (164 mmHg) also indirectly indicates the exercise intensity in our running protocol. It should be noted that the time between the treadmill run and the blood pressure assessment took approximately 4-minute. Since cardiovascular function (heart rate and blood pressure) is rapidly adapting [ 33], we assume that the actual systolic blood pressure was higher than what we observed. Recorded blood pressures at the pre-measurements were higher than what are considered normal (systolic: <120 mmHg; diastolic: <80 mmHg) [ 34]. Since having the rest period (>10-minute) for cardiovascular stability prior to the treadmill jog or run, influence of the fingerstick on the autonomic nervous system could be a good candidate to explain why our subjects showed higher blood pressure values. We acknowledge that this is a methodological weakness; thus, in future studies, researchers should be aware of this effect on the measurement order. Our study has limitations that should be noted when applying the results. First, our interventions, treadmill jogging and running, were not randomised: all subjects performed jogging first followed by running. A study design with prospective randomisation is considered as gold standard for examining cause-and-effect relationships. Randomisation is particularly important for providing the intervention effects (e.g., benefit or harm) [ 35]. Our study purpose was to observe the responses, not to determine the superiority between jogging and running. Additionally, two-weeks was sufficient for potential learning effect from the first experiment while many sports science studies [ 36, 37] with a crossover design had a washout period of a week. Therefore, we believe that the results were minimally affected by the order effects. We also acknowledge that not all subjects (n=5) visited the laboratory at the same time of the day due to scheduling conflicts. Our subjects were recreationally active, healthy male college students. Untrained or well-trained individuals at different ages may respond differently. Care should be taken when referencing our data to pathological populations such as hypertension or diabetes. Recorded mean relative humidities in the laboratory during the data collection sessions were different (jog: 44%; run: 71%). Since a short bout of exercise at a high-intensity level is not compromised by the relative humidity [ 38], it is unlikely that the outcomes were affected by the differences.
CONCLUSION
We compared physiological and psychological responses following 10-minute treadmill jogging and running. Subjects who ran showed higher values in heart rate (187 vs. 158 bpm), blood lactate concentration (11.4 vs. 3.8 mmol/L), systolic blood pressure (164 vs. 147 mmHg), and fatigue perception (5.0 vs. 2.7 cm) compared with those who jogged. The temperature increases in the gastrocnemius muscle in both conditions were similar (jog: 1.7°C; run: 1.5°C). Our data could be used as basis for planning and implementing warm-up protocols for recreationally active young adults.
REFERENCES
1. Segers V, Lenoir M, Aerts P, De Clercq D. Kinematics of the transition between walking and running when gradually changing speed. Gait Posture. 2007;26(3):349-61.   2. Gazendam MG, Hof AL. Averaged EMG profiles in jogging and running at different speeds. Gait Posture. 2007;25(4):604-14.   3. Ho IJ, Hou YY, Yang CH, Wu WL, Chen SK, et al. Comparison of plantar pressure distribution between different speed and incline during treadmill jogging. J Sports Sci Med. 2010;9(1):154-60.   4. Diaz KM, Krupka DJ, Chang MJ, Peacock J, Ma Y, et al. Fitbit Ⓡ: an ac-curate and reliable device for wireless physical activity tracking. Int J Cardiol. 2015;185:138-40.   5. Gonçalves B, Esteves P, Folgado H, Ric A, Torrents C, et al. Effects of pitch area-restrictions on tactical behavior, physical, and physiological performances in soccer large-sided games. J Strength Cond Res. 2017;31(9):2398-408.   7. Kim MS, Park J. Immediate effects of an inverted body position on energy expenditure and blood lactate removal after intense running. Appl Sci. 2020;10(19):6645.  8. Van den Tillaar R, Vatten T, von Heimburg E. Effects of short or long warm-up on intermediate running performance. J Strength Cond Res. 2017;31(1):37-44.   9. Tsurubami R, Oba K, Samukawa M, Takizawa K, Chiba I, et al. Warm-up intensity and time course effects on jump performance. J Sports Sci Med. 2020;19(4):714-20.   10. Min KE, Lee Y, Park J. Changes in spinal-reflex excitability during stat-ic stretch and/or explosive contraction. Appl Sci. 2021;11(6):2830.  11. Silva LM, Neiva HP, Marques MC, Izquierdo M, Marinho DA. Effects of warm-up, post-warm-up, and re-warm-up strategies on explosive efforts in team sports: a systematic review. Sports Med. 2018;48(10):2285-99.   12. Brajkovic D, Ducharme MB, Webb P, Reardon FD, Kenny GP. Insulation disks on the skin to estimate muscle temperature. Eur J Appl Physiol. 2006;97(6):761-5.   13. Kikuchi K, Asano M, Tagami H, Kato M, Aiba S. Comparison of the measuring efficacy of transepidermal water loss of a reasonably priced, portable closed-chamber system device H4500 with that of rather ex-pensive, conventional devices such as Tewameter Ⓡ and Vapometer Ⓡ
. Skin Res Technol. 2017;23(4):597-601.   15. Colella TJ, Tahsinul A, Gatto H, Oh P, Myers MG. Antecedent rest may not be necessary for automated office blood pressure at lower treatment targets. J Clin Hypertens. 2018;20(8):1160-4.  16. Cohen J. The effect size index: d. Statistical power analysis for the behavioral sciences. Abingdon-on-Thames: Routledge Academic 1988.
17. Borresen J, Lambert MI. Autonomic control of heart rate during and after exercise. Sports Med. 2008;38(8):633-46.   18. Cottin F, Médigue C, Leprêtre PM, Papelier Y, Koralsztein JP, et al. Heart rate variability during exercise performed below and above ven-tilatory threshold. Med Sci Sports Exerc. 2004;36(4):594-600.   20. De Ruiter C, De Haan A. Temperature effect on the force/velocity relationship of the fresh and fatigued human adductor pollicis muscle. Pflugers Arch. 2000;440(1):163-70.   21. Racinais S, Oksa J. Temperature and neuromuscular function. Scand J Med Sci Sports. 2010;20(3):1-18.  23. Drinkwater E. Effects of peripheral cooling on characteristics of local muscle. Thermoregulation and Human Performance. 2008;53:74-88.  24. Drust B, Rasmussen P, Mohr M, Nielsen B, Nybo L. Elevations in core and muscle temperature impairs repeated sprint performance. Acta Physiol Scand. 2005;183(2):181-90.   25. Racinais S, Cocking S, Périard JD. Sports and environmental temperature: from warming-up to heating-up. Temperature. 2017;4(3):227-57.  26. Tillin NA, Bishop D. Factors modulating postactivation potentiation and its effect on performance of subsequent explosive activities. Sports Med. 2009;39(2):147-66.   27. Dayal A, Schrötter K, Pan Y, Föhr K, Melzer W, et al. The Ca 2+ influx through the mammalian skeletal muscle dihydropyridine receptor is irrelevant for muscle performance. Nat Commun. 2017;8(1):1-14.   30. Fell J, Rayfield J, Gulbin J, Gaffney P. Evaluation of the Accusport Ⓡ lactate analyser. Int J Sports Med. 1998;19(3):199-204.  31. Sharman J, LaGerche A. Exercise blood pressure: clinical relevance and correct measurement. J Hum Hypertens. 2015;29(6):351-8.   32. Kenny WL, Wilmore JH, Costill DL. Physiology of sport and exercise. 6th ed., Champaign: Human Kinetics 2021;pp. 189-92).
33. Evans DL, Young LE. Cardiac responses to exercise and training. Car-diology of the Horse E-Book. 2011;35.  35. Higgins J, Thomas J, Chandler J, Cumpston M, Li T, et al. Cochrane handbook for systematic reviews of interventions. Wiley 2021.
37. Heyman EGB, Mertens I, Meeusen R. Effects of four recovery methods on repeated maximal rock climbing performance. Med Sci Sports Exerc. 2009;41(6):1303-10.   38. Backx K, Mc Naughton L, Crickmore L, Palmer G, Carlisle A. Effects of differing heat and humidity on the performance and recovery from multiple high intensity, intermittent exercise bouts. Int J Sports Med. 2000;21(06):400-5.  
|
|






